How to Draw Molecular Orbital Diagram of O2
Molecular Orbital (MO) Theory is the last theory pertaining to the bonding between molecules. In contrast to VSEPR and valence bond theory which draw bonding in terms of atomic orbitals, molecular orbital theory visualizes bonding in relation to molecular orbitals, which are orbitals that surround the entire molecule. The purpose of MO theory is to make full in the gap for some behavior that cannot be explained by VSEPR and Valence-Bond Theory. Unfortunately, MO Theory can be the well-nigh hard to empathize and visualize, which is why nosotros covered the other ii first.
As we mentioned in before posts, the manner nosotros determined the shape of orbitals is through Schrodinger'south wave equation. Information technology turns out that, co-ordinate to the wave equation, orbitals tin exist one of two states. These states are often written as `+` and `-`, or drawn as different colors.
For instance, the p-orbitals are ordinarily drawn with the two regions equally different colors. We'll telephone call this the "sign" of the orbital.

s-orbitals are usually written as having one of 2 colors. What happens when s-orbitals interact? There are two possibilities: the first of which being that ii s-orbitals of the same sign interact, and the second of which beingness that s-orbitals of the opposite sign interact. The results of these 2 are shown in the images below:
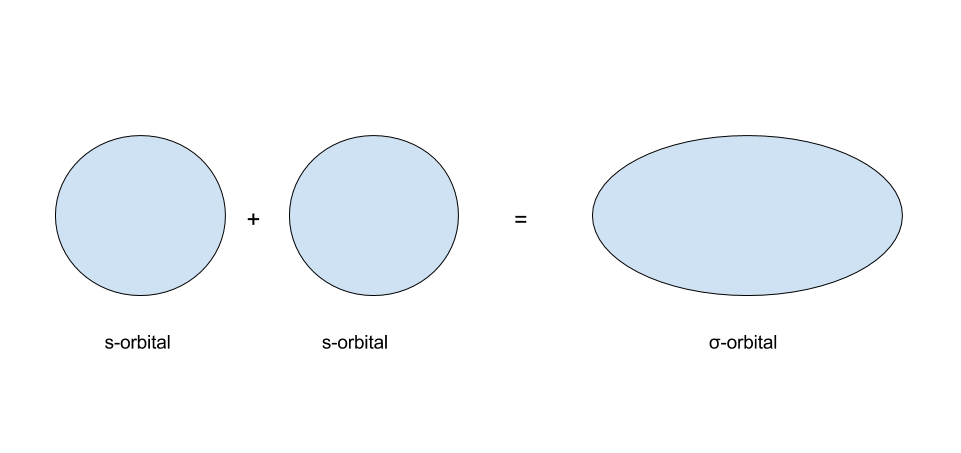
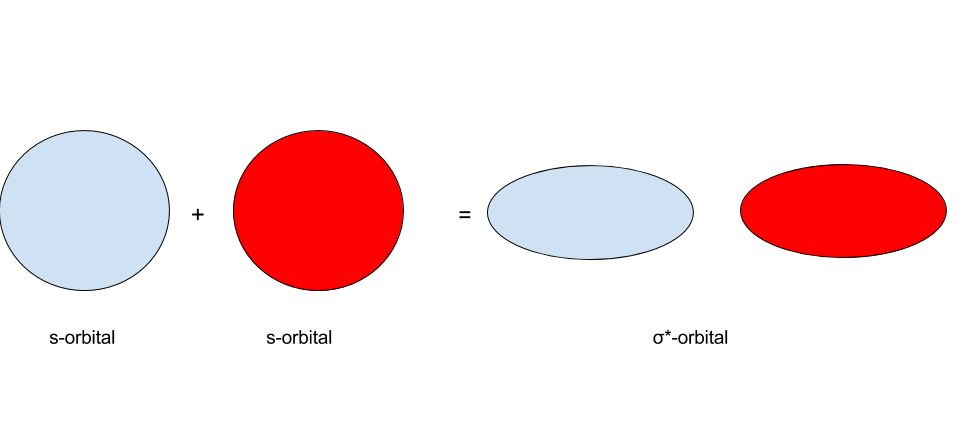
In MO theory, when two orbitals interact, they form a set up of molecular orbitals. When two due south-orbitals of the same sign collaborate, they grade a `sigma"-bonding"` orbital. When 2 s-orbitals of the opposite sign interact, they grade a `sigma"-antibonding"` orbital. Whenever 2 orbitals interact to form molecular orbitals, they form a set of two molecular orbitals: 1 bonding orbital and one antibonding orbital. This ties into the earlier concept that the number of orbitals must remain conserved.
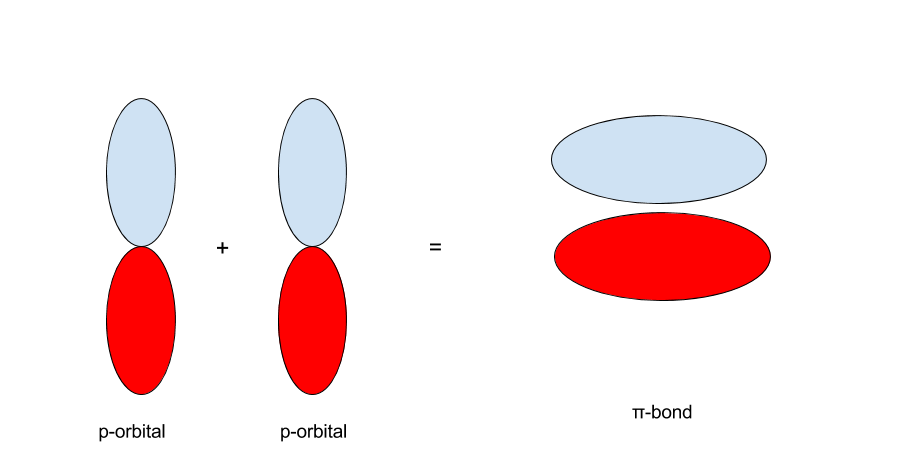
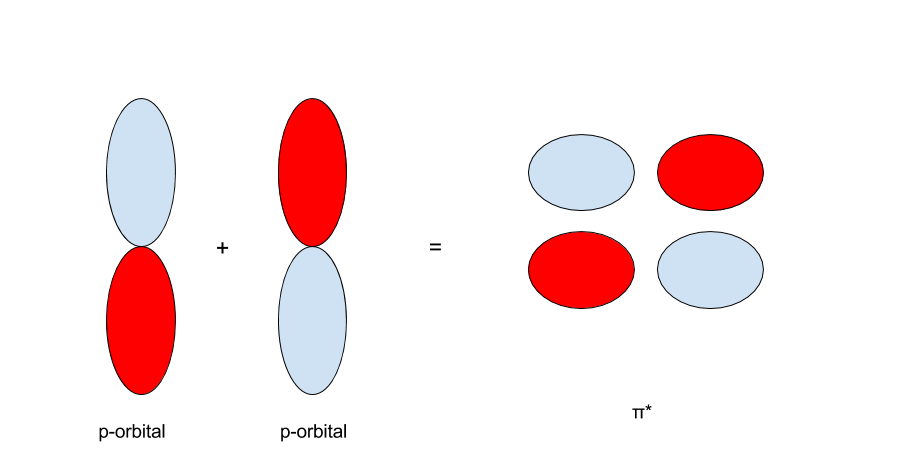
The same principle applies to the p-orbitals. With two p-orbitals along the same orientation, there are two possibilities. The first is that the p-orbitals interact such that the red regions collaborate with the red and the blue with the blue. The 2nd is that the cherry-red interacts with the blue and the blueish with the red.
When p-orbitals with the aforementioned sign and orientation interact, they grade a `pi`-bond:
On the other hand, when the p-orbitals are oriented with opposite signs, they form a `pi^"*"`- bond, pronounced "`pi`-antibonding."
The proper note is that molecular orbitals are written merely past the kind of bail that the orbital creates. An anti-bonding orbital is written as the bail with the star superscripted onto it. For example:
`sigma`-bonding = `sigma`
`sigma`-anti-bonding = `sigma^"*"`
`pi`-bonding = `pi`
`pi`-anti-bonding = `pi^"*"`
MO theory explains when single and double bonds volition be formed. A `sigma"-bond"` corresponds to a single bond and a `pi"-bail"` corresponds to a double bail. This explains where double bonds come up from: double bonds are formed through electrons in the p-orbital. This means that single bonds are formed through s-orbitals whereas double bonds are formed from p-orbitals. In a triple bond for instance, the kickoff bond comes from the southward-orbital, the 2nd from the p-orbitals, and the third also from the p-orbitals.
The concept of an anti-bonding orbital is new, since VSEPR/valence bond do not consider antibonding orbitals. In MO theory, electrons in bonding orbitals promote bonding whereas electrons in anti-bonding orbitals weaken bonds. We'll see the awarding of this when we go over MO diagrams later in this post.
There's a lot of data in this section that probably didn't brand sense. After all, MO theory is one of the almost complicated sections covered in general chemical science. I recommend reading it over a few times until the following concepts are understood:
1. When two atomic orbitals collaborate, they form a bonding orbital and an anti-bonding orbital.
2. Electrons in bonding orbitals will strengthen the bonds of the molecule. Electrons in an anti-bonding orbital volition weaken the bonds of the molecule.
three. Single bonds come from electrons in s-orbitals. Any additional bonds come up from electrons in p-orbitals.
In this department, we're going to learn how to depict and utilize MO diagrams. MO diagrams allow us to decide diverse properties that cannot be determined via. VSEPR.
MO diagrams expect like this:
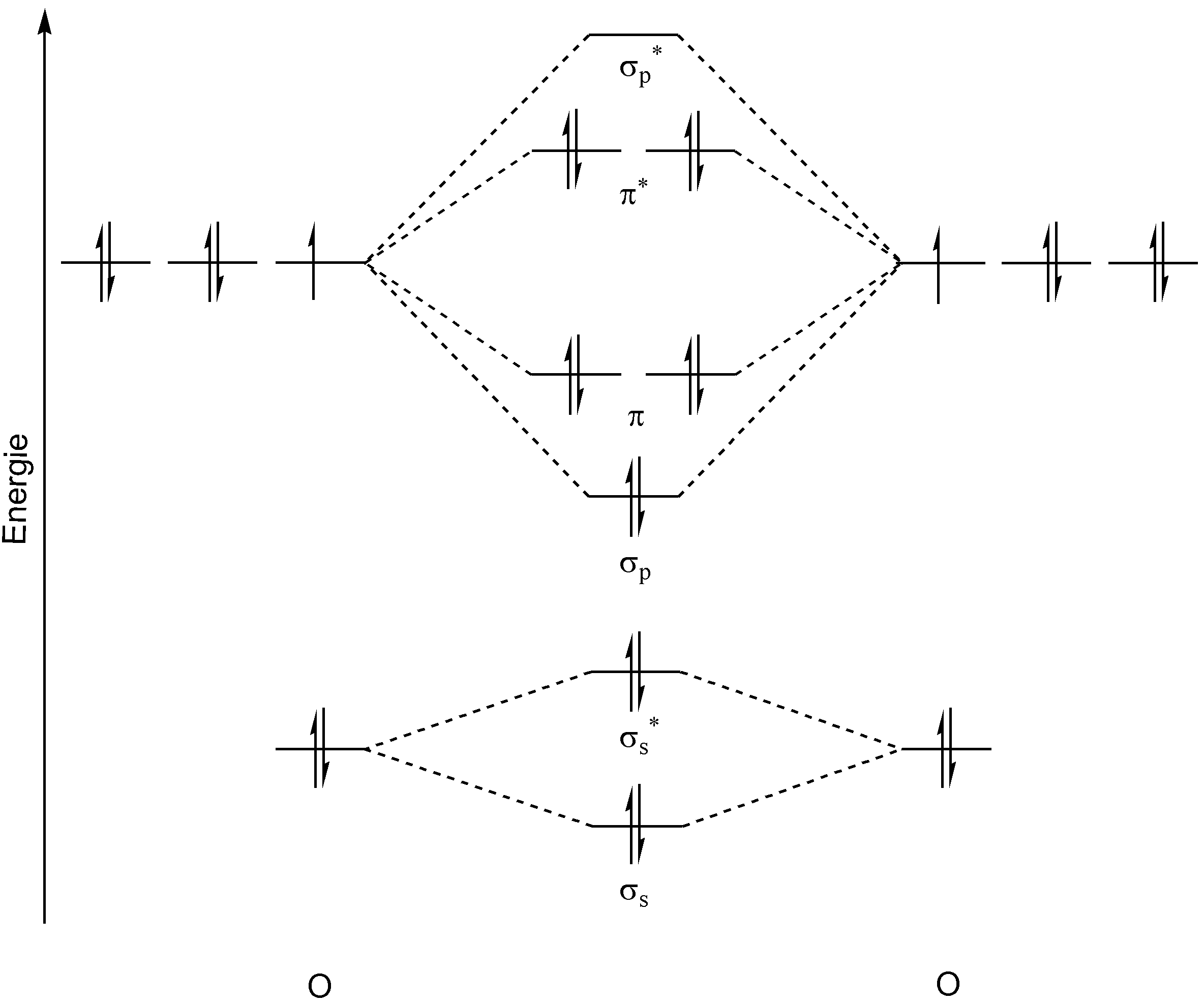
They're not every bit intimidating equally they may seem. For now, we're but covering homonuclear MO diagrams which involve the diatomic molecules composed of the same element. The elements nosotros're roofing will exist the ones in period 2, from `Li` to `F`. There are 2 MO diagrams nosotros demand to acquire for these elements. The outset is for all molecules except for `O_2` and `F_2`. The second is for just `O_2` and `F_2`.
The diagram for all molecules except for `O_2` and `F_2` is the following:
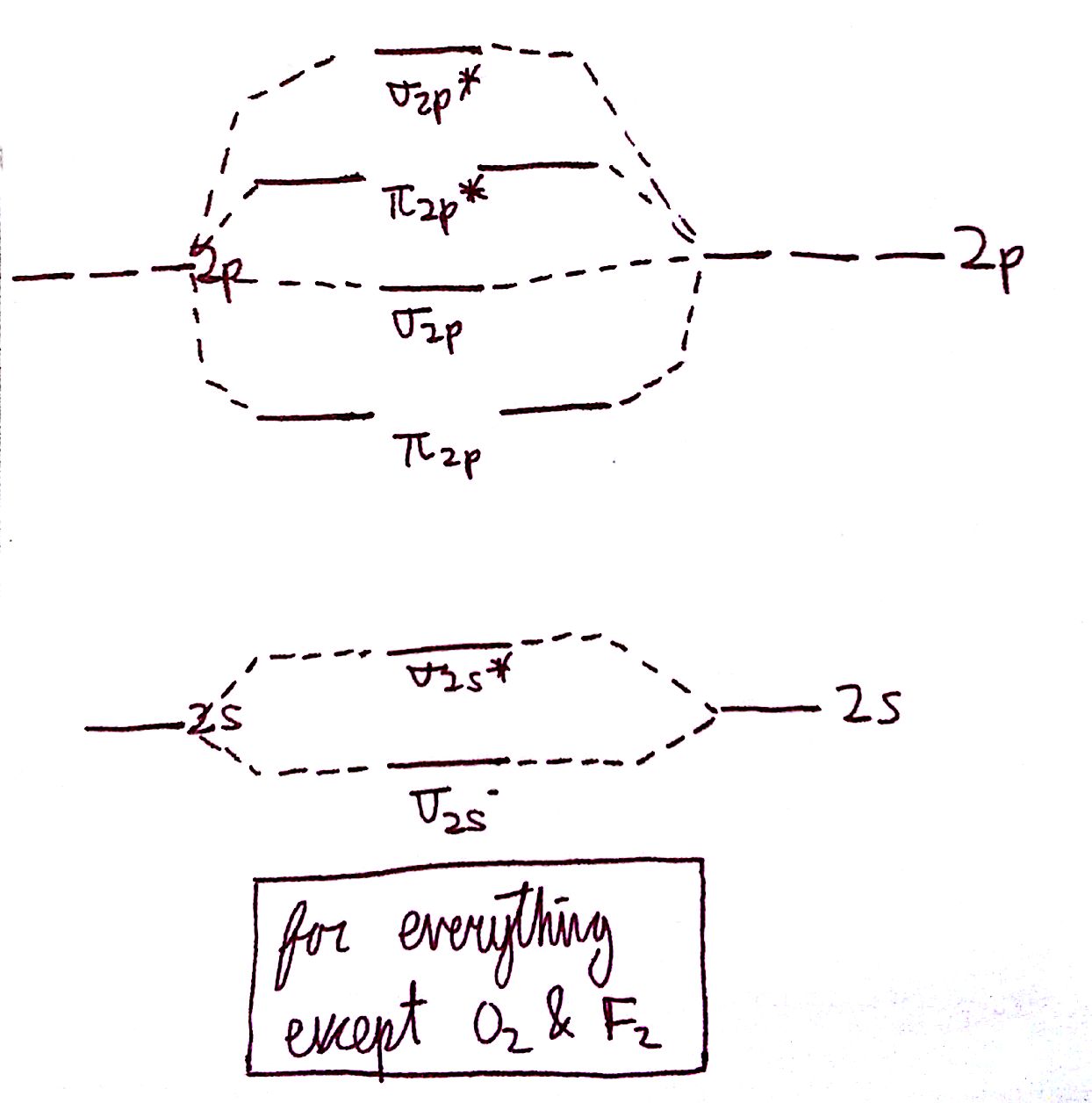
The diagram for simply `O_2` and `F_2` is here:
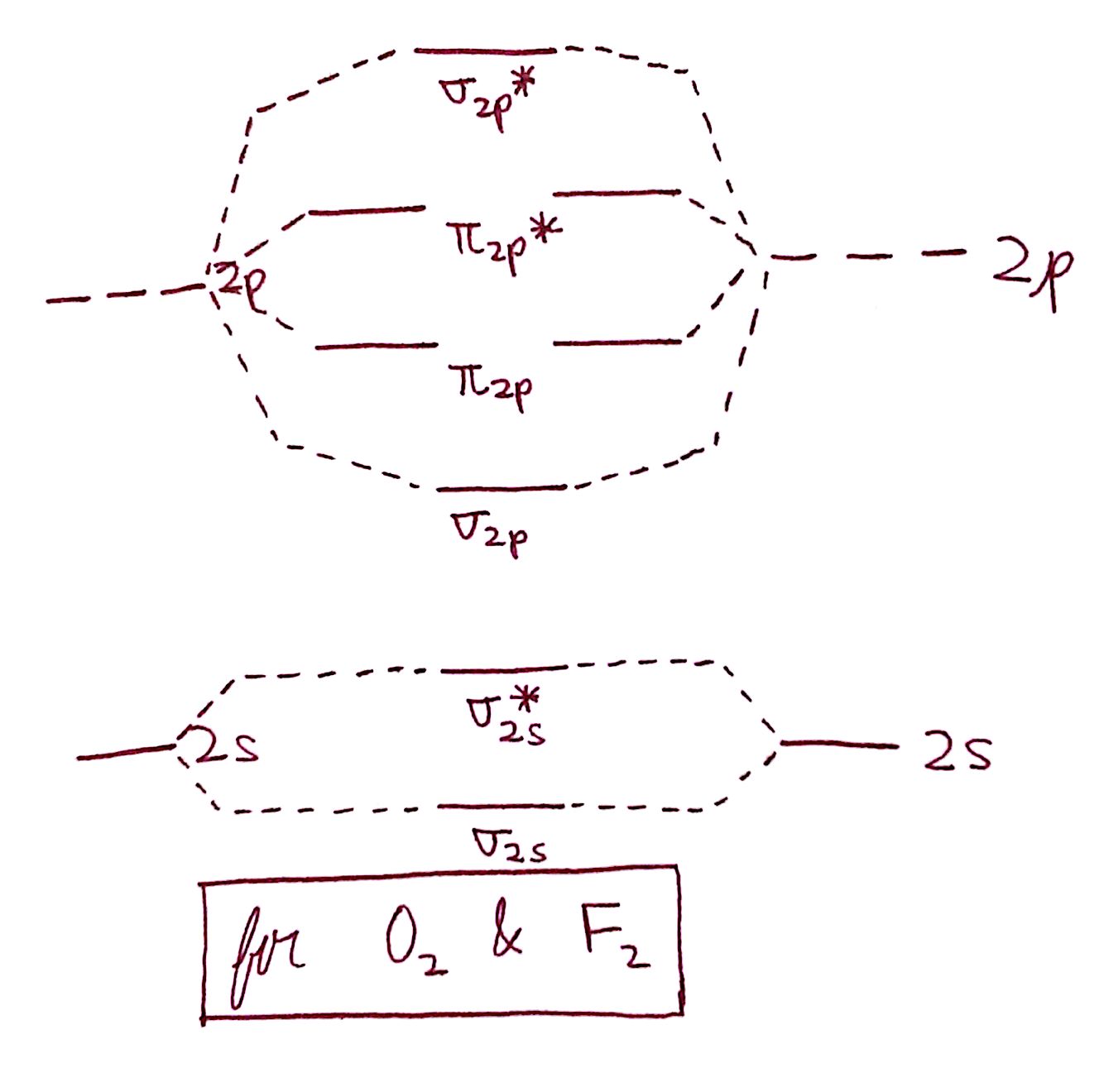
Do you notice the difference? In the diagram for `O_2` and `F_2`, the `sigma_("2p")` and `pi_("2p")` are reversed. This very slight departure becomes profoundly of import when it comes to the behavior of `O_2` and `F_2`.
Now let's acquire how to fill out the diagrams. We can break it down into several smaller steps.
1. Make up one's mind the total number of valence electrons.
The molecules we'll be dealing with in MO diagrams are all homonuclear moleculars east.g `B_2`, `O_2`, etc. The total number of valence electrons is twice the number that the atomic species would have, since there are now ii of the atoms in the molecule.
For example, `O` has `6` valence electrons. `O_2` is comprised of two `O` atoms, then it has `2(half-dozen)=12` valence electrons.
two. Determine the number of electrons in the `s` and `p` orbitals.
Recall that in the `north=2` energy level, there is ane s-orbital and iii p-orbitals. For whatever molecule, decide the number of electrons in both the south and p-orbitals.
For example, `B` has `2` electrons in the `2s` orbital and `i` electron in the `2p` orbitals. `F` has `2` electrons in the `2s` free energy level and `v` in the `2p` orbitals. This is an application of electron configuration; if this is unfamiliar, cheque out this mail service:
iii. Fill in the electrons into the molecular orbitals in the correct MO diagram.
Make sure you're using the correct MO diagram! When filling in molecular orbitals, all of the principles for filling in orbitals (Hund's Rule, Pauli Exclusion Principle, Aufbau Principle), still employ! Here's how they apply:
ane. Pauli Exclusion Principle: each molecular orbital tin can accomodate two electrons.
two. Aufbau Principle: electrons will e'er make full the orbitals from bottom to superlative. This means that we always offset with the `2s` orbitals and fill upward.
three. Hund'due south Rule: orbitals on the same energy level volition fill singly before doubly. This applies primarily to the `pi` and `pi^"*"` orbitals, where one electron will go into each orbital before filling in the second.
Only like with electron configuration, MO diagrams will always fill the same manner! Once you understand the full general design, none of the MO diagrams covered should be a problem.
In summary: fill in the orbitals from the bottom up. Each orbital tin concur two electrons, so orbitals lower should always fill upward before the orbitals higher up are filled. In the case of the 2 `pi` orbitals, one electron goes into each orbital before two get into either. The MO diagram is complete when all of the valence electrons are used.
Let's demonstrate these principles with a couple problems.
#1. Draw the MO diagram for `B_2`.
Starting time footstep is to make up one's mind which MO diagram we're using. In this instance, nosotros're using the standard 1.
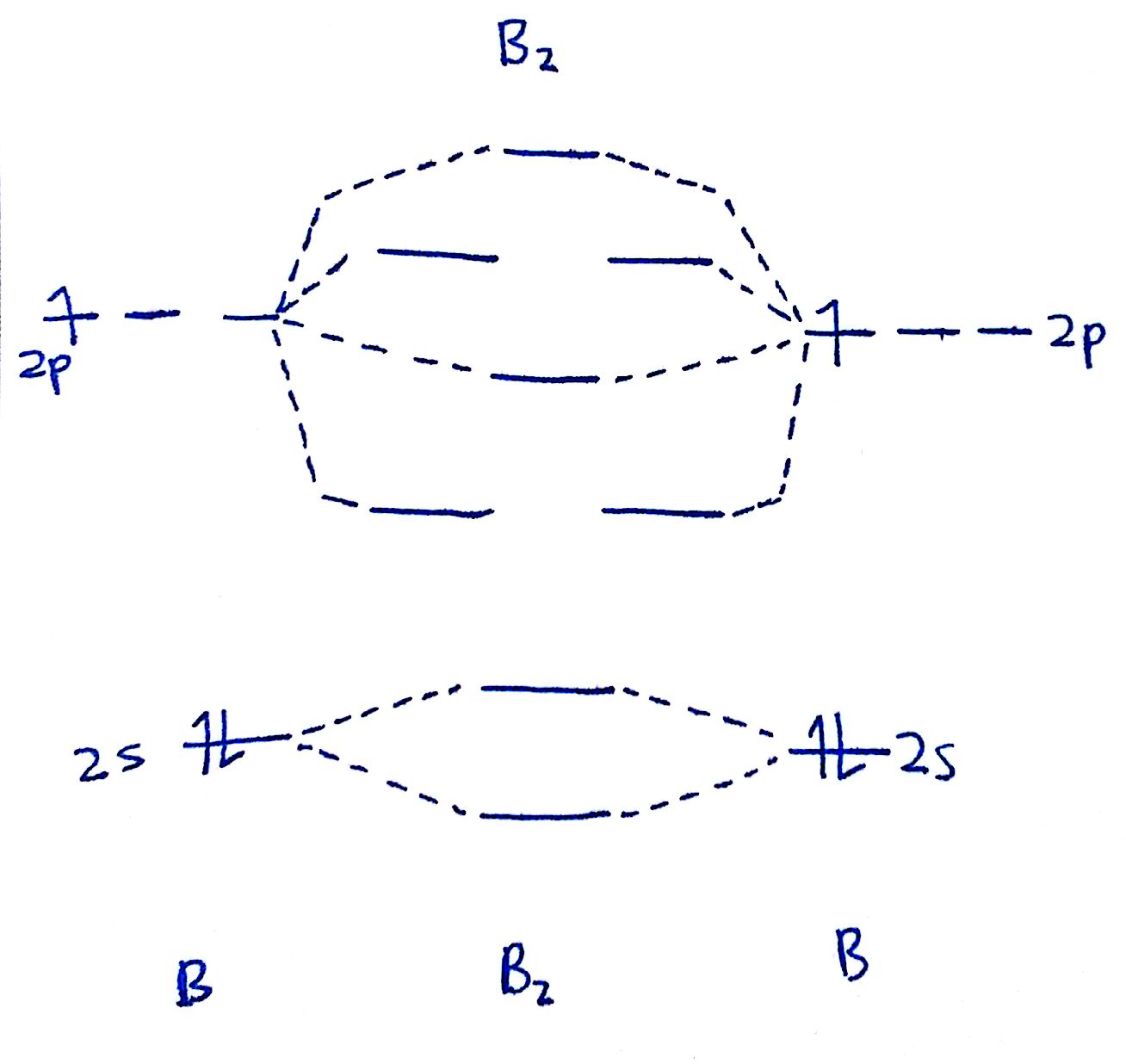
Draw out the MO diagram and label in the valence electrons. Boron has 2 electrons in the `2s` orbitals and 1 electron in the `2p` orbital.
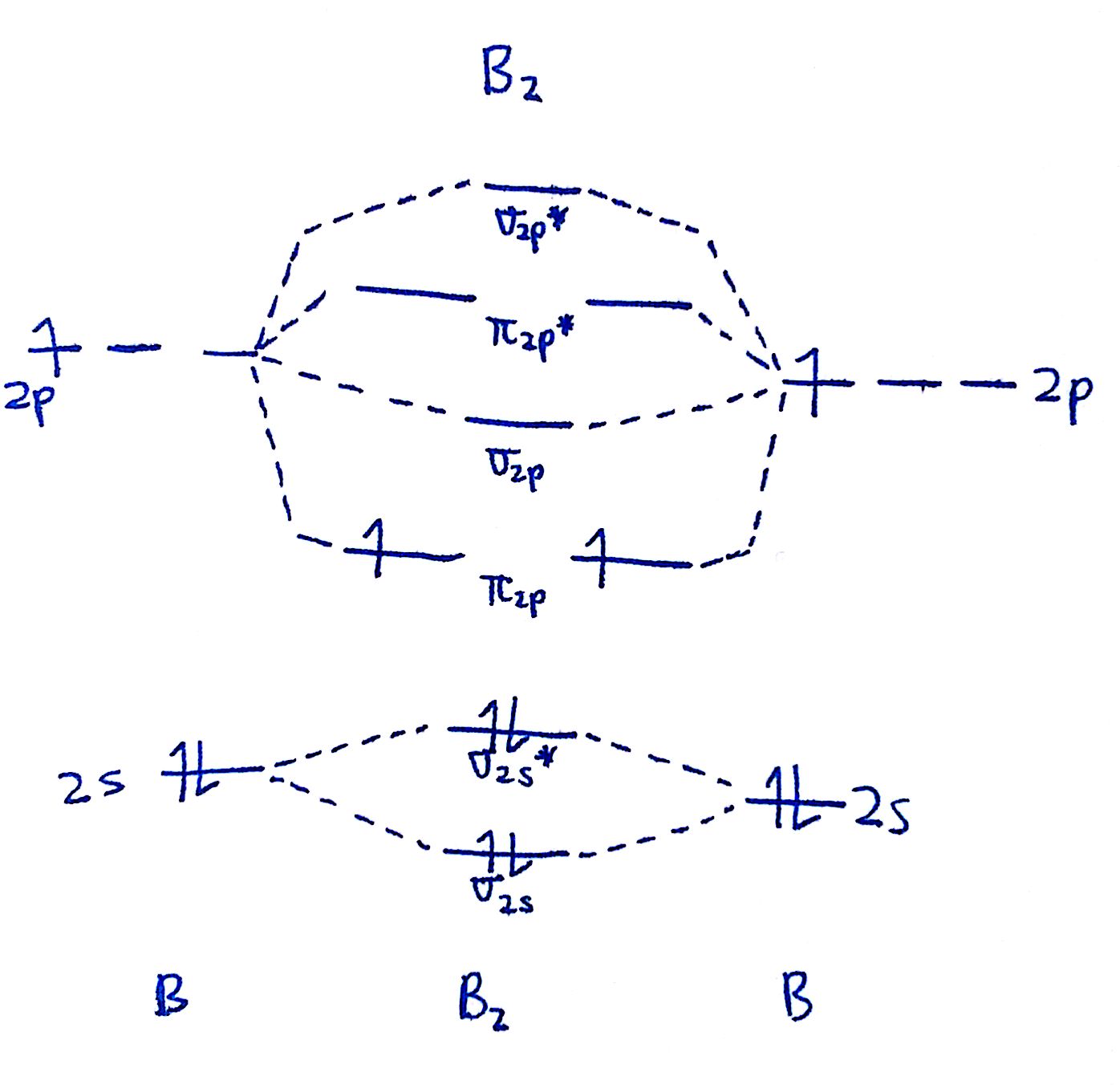
That's it for the MO diagram of `B_2`! To check, count how many electrons there are in full. `B_2` has `2(3)=six` valence electrons. The MO diagram has `six` electrons likewise. Notice that the last ii electrons go into two dissever `pi` orbitals instead of filling 2 electrons into ane orbital. This is in accordance to Hund's Rule.
#ii. Draw the MO diagram for `O_2`
Since nosotros're doing the MO diagram for `O_2`, we have to employ the `O_2` MO diagram which features flipped `pi_"2p"` and `sigma_"2p"` orbitals. Fill out the valence electrons.
Now, make full in the electrons from the bottom up. `2` electrons go into the `sigma_"2s"`, `2` into the `sigma_"2s"^"*"`, `2` into the `sigma_"2p"`, `four` into the `pi_"2p"`, and `2` into the `pi_"2p"^"*"`. The resulting diagram should await like this.
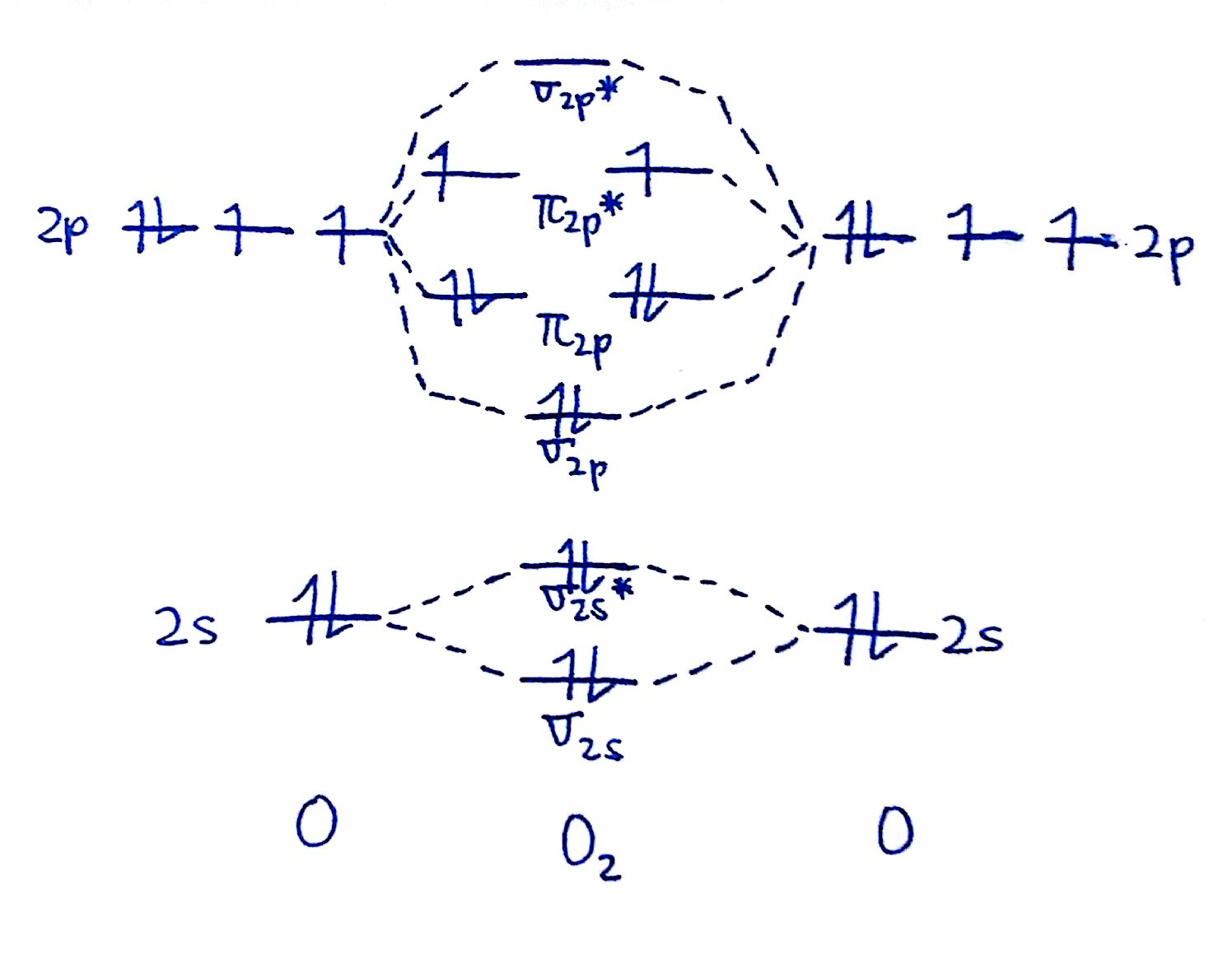
#3. Draw the MO diagram for `O_2^+`
This is a fleck of a curveball, merely a perfectly valid problem. Recall that a cation indicates a loss of `i` electron. `O_2^+` is just the ionized form of `O_2`; that is, it'due south `O_2` with `1` missing electron.
The MO diagram will exist the same as the MO diagram of `O_2`, except with `1` less electron. You can either depict the `O_2` diagram and remove `1` electron, or just depict the `O_2^+` diagram. The diagram will end up as such:
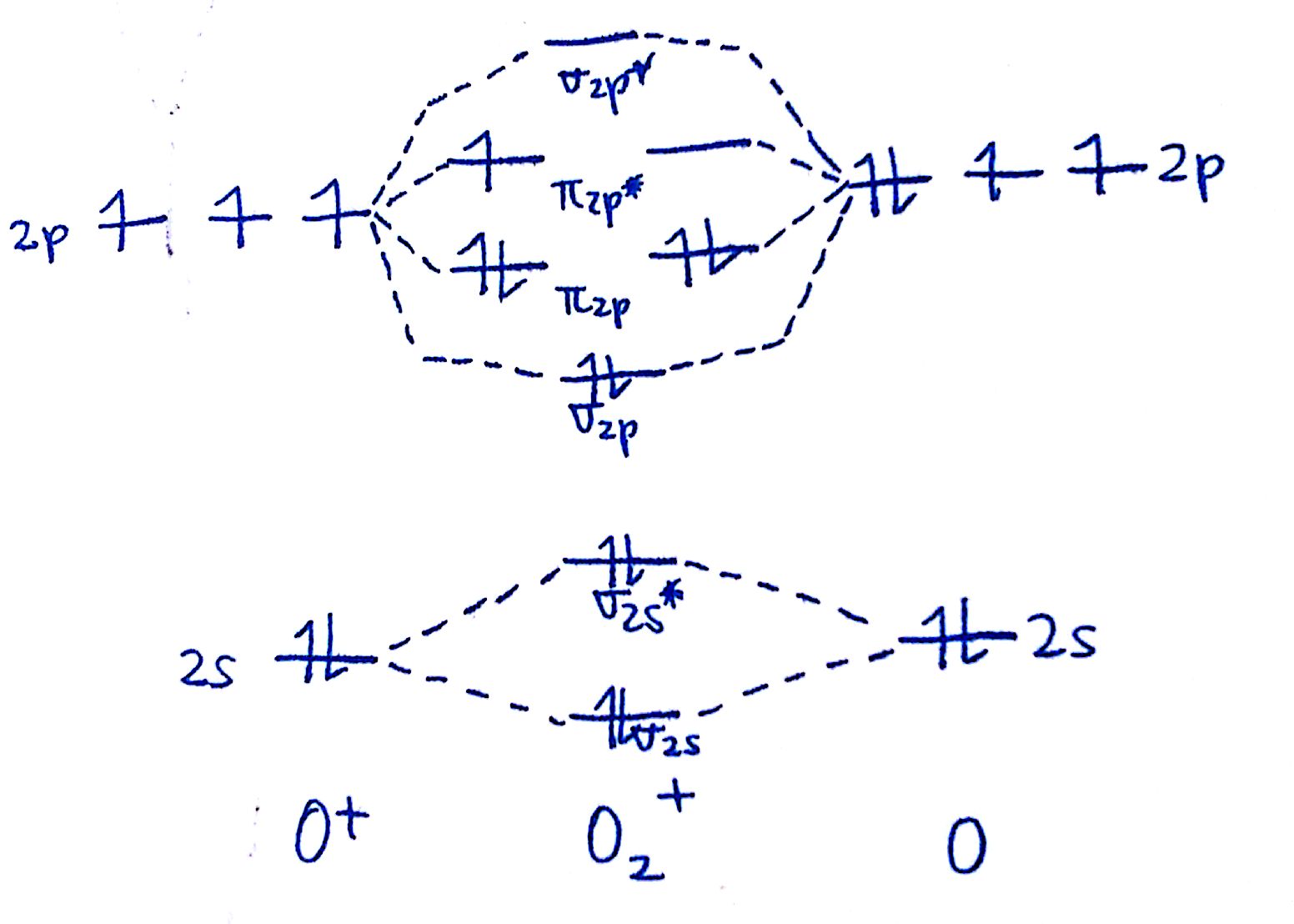
Observe the consequence that this has on the overall bonds. Think from earlier that electrons in bonding orbitals will stregnthen bonds whereas electrons in antibonding orbitals will weaken bonds. Past removing an electron from an antibonding orbital, the `O-O` bond is actually getting stronger! This brings us to the concept of the bond order.
We can now expand on the concept of the bond order. In an earlier section, we learned that the bond order is defined equally such:
`"Bond Order"=("Number of Bonds")/("Number of Bonded Groups")`
The bond order tells us the boilerplate number of bonds between the bonded atoms. In a diatomic molecule such as `O_2`, the bond lodge merely tells the number of bonds between the 2 atoms.
The bond order can be interpreted from MO diagrams using the following formula:
`"Bond Order" = 1/two [("Bonding "e^-)-("Antibonding " e^-)]`
The 2 formulas for bond society tell us the same information. The value in the bail order from MO diagrams is that nosotros tin now decide the number of bonds in betwixt atoms that we otherwise would not be able to.
For case, here are the MO diagrams for `"Ne"_2` and `O_2^-`. We know that `"Ne"_2` should not exist seeing as `"Ne"` is a noble gas. What can nosotros say about `O_2^-`?
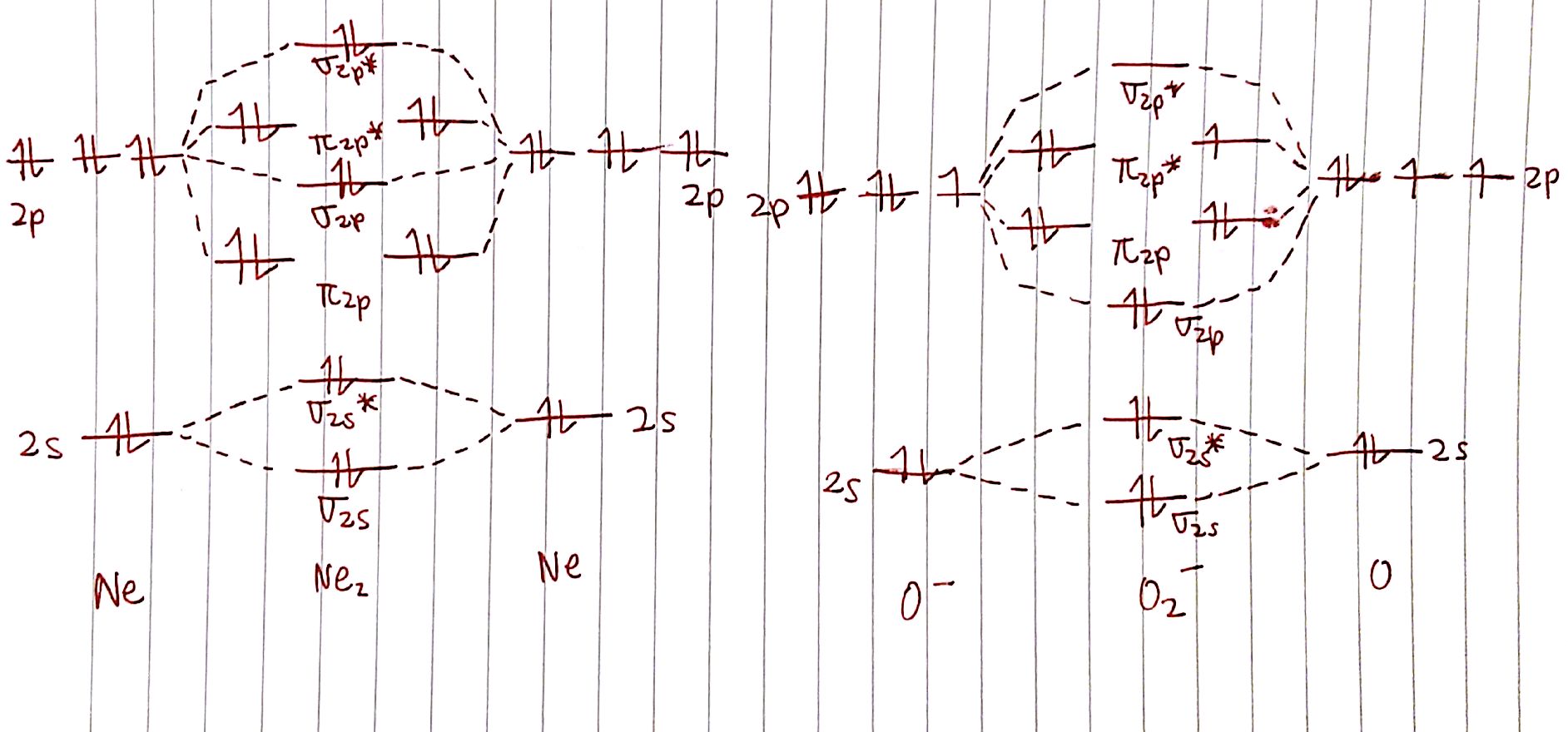
Apologies for the binder paper, I ran out of printer paper.
Let's calculate the bond social club of `"Ne"_2`. There are `eight` electrons in bonding orbitals and `eight` electrons in antibonding orbitals. The bond order is therefore:
`BO=one/2 (8-viii)=0`
This is in line with how we expect noble gases to behave! What about `O_2^-`?
`BO=1/2(8-5)=iii/two`
Normal `O_2` has a bond gild of two. This means that, going from `O_2` to `O_2^-`, the bonds between the `O` atoms weakens! If we ionize `O_2` into `O_2^+`, the bail lodge becomes `i/2 (8-3)=5/ii` , which means that the bonds are condign stronger!
With MO diagrams, we tin can predict the number of bonds in diatomic molecules. For case, here's the MO diagram for `N_2`. We know from the Lewis structure that `N_2` has a triple bond. This means that the bond order of `N_2` should be `three`.
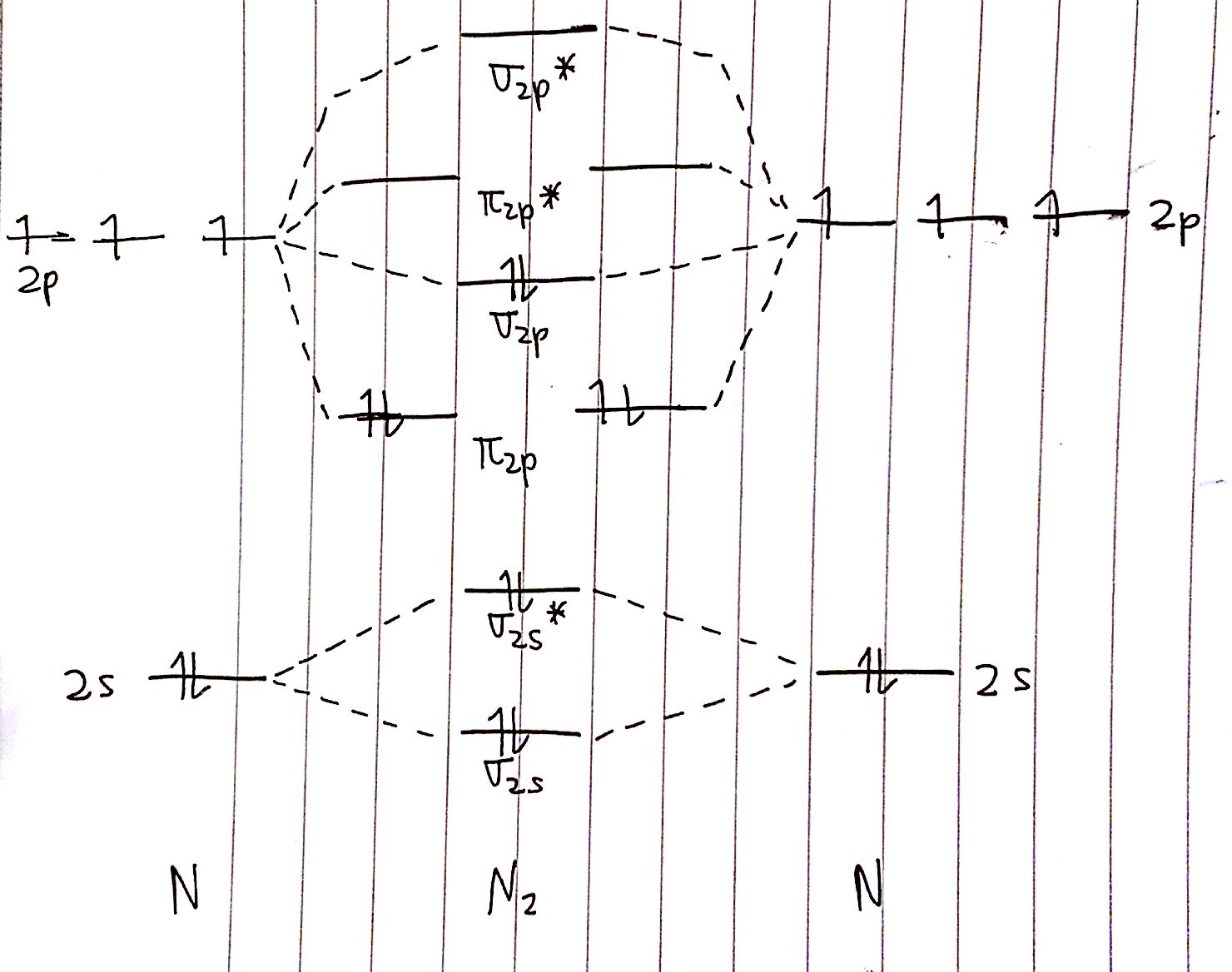
The bail order is calculated as follows:
`BO=ane/2 (8-ii)=3`
This is exactly what we expected!
The magnetic properties of a molecule tin exist determined through the molecule's MO diagram. Magnetism results from unpaired electrons.
If we draw out `O_2`'southward Lewis dot construction, we'll detect no unpaired electrons:
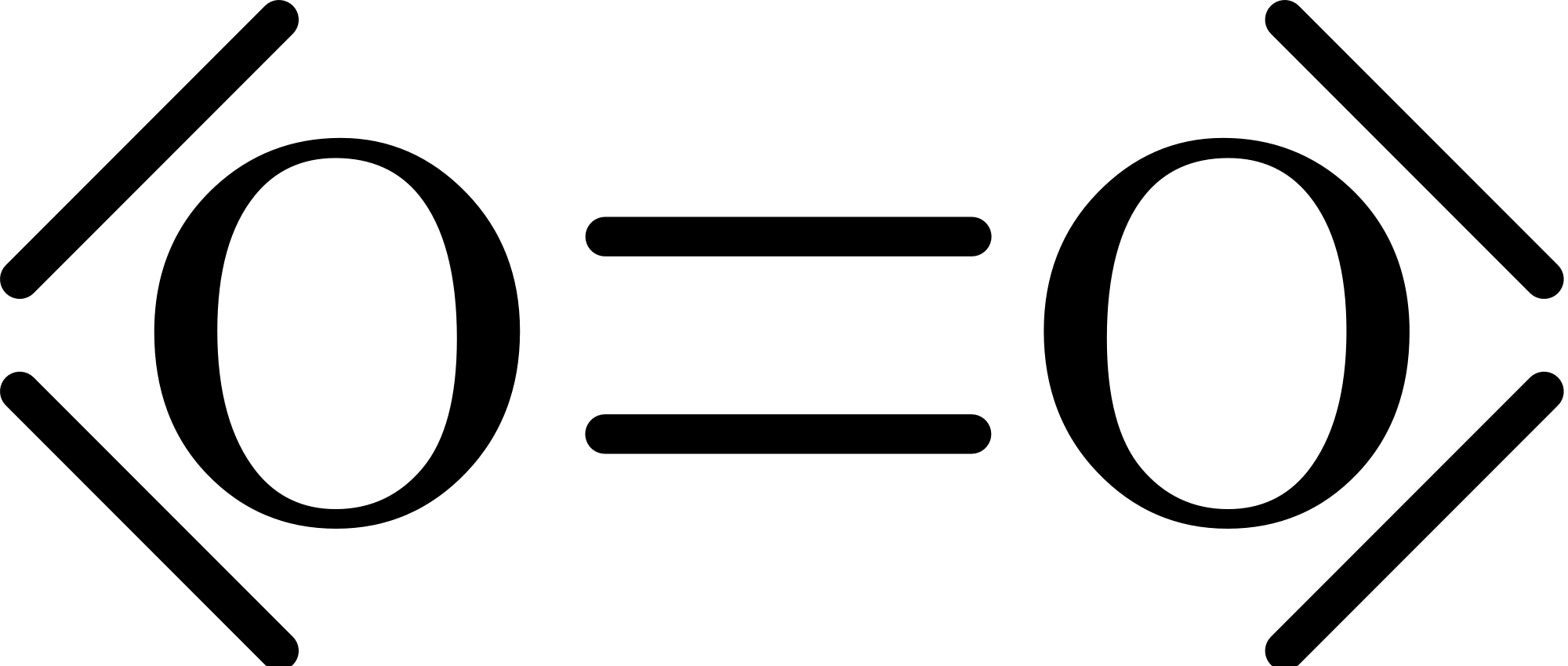
However, from experiments we know that `O_2` gas is actually magnetic. We can encounter that in the following .gif. When oxygen gas is poured in betwixt ii magnets, the gas is attracted to the magnets.

Why is it that, if magnetism results from unpaired electrons, that `O_2` is magnetic? To answer this, we have to examine the MO diagram of `O_2`.
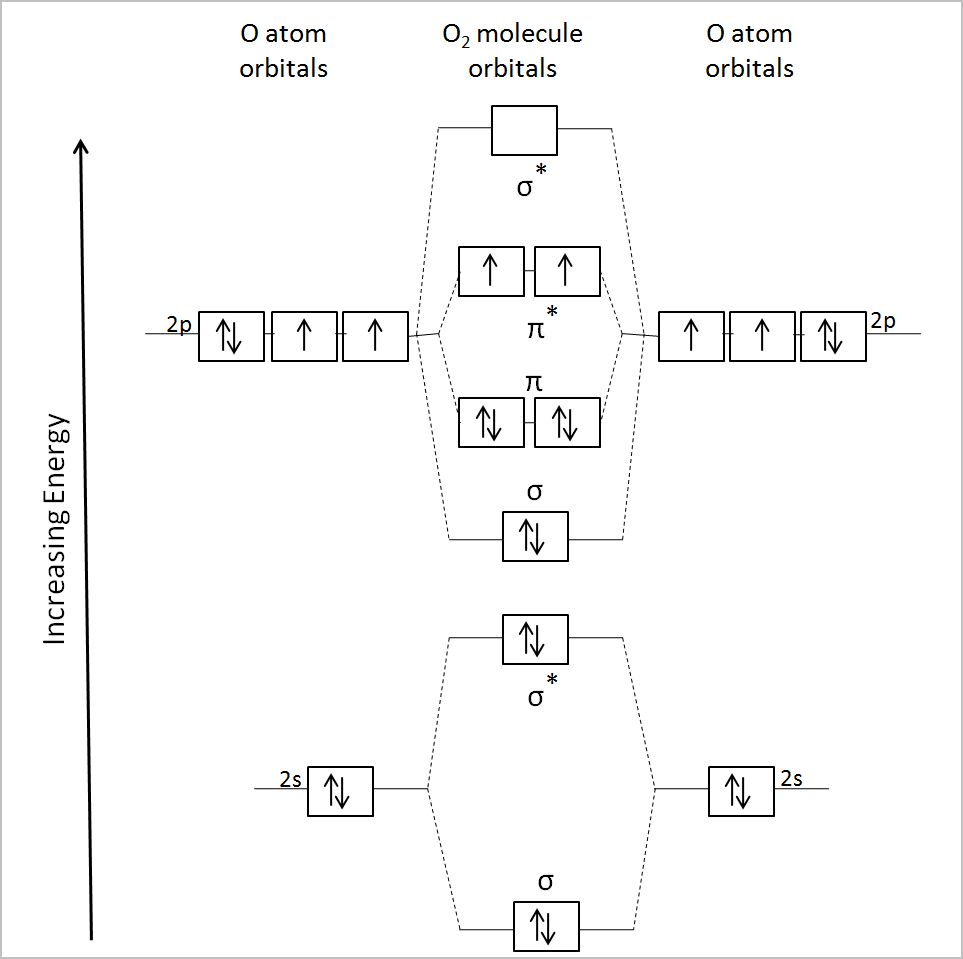
In the `pi^"*"` orbitals, the ii electrons are unpaired. This is why `O_2` is magnetic!
We can classify magnetic properties into two different categories:
1. Paramagnetic: when unpaired electrons exist.
2. Diamagnetic: no unpaired electrons be.
From these definitions, we can classify `O_(2(g))` every bit having paramagnetic behavior. Diamagnetic molecules are molecules that exhibit no magnetic properties due to the lack of unpaired electrons. `N_2` gas, for example, is diamagnetic.
one. Co-ordinate to MO theory, when ii diminutive orbitals interact, they class one bonding orbital and one antibonding orbital.
ii. Electrons in bonding orbitals strengthen bonds whereas electrons in antibonding orbitals weaken bonds.
3. `sigma` - bonds are formed through the interaction of south-orbitals. `sigma` - bonds are equivalent to single bonds.
4. `pi` - bonds are formed through interaction of p-orbitals. `pi` - bonds are equivalent to double bonds.
5. Whenever a multiple bond (double, triple) exists, the commencement bond is a `sigma` - bond and the residuum are `pi` - bonds. For example, a double bail consists of `one sigma`- bail and `1 pi` - bond. A triple bond consists of `i sigma` - bond and `ii pi` - bonds.
6. MO diagrams permit us to view the specific configuration of valence electrons in their molecular orbitals.
7. Magnetism is a phenomena due to unpaired electrons. Species with unpaired electrons are paramagnetic whereas species with all electrons paired are diamagnetic.
one. Color of organic compounds
The color of many organic compounds comes from their conjugated systems. A conjugated arrangement is a system in which the bonds alternate from single to double to single. For example:
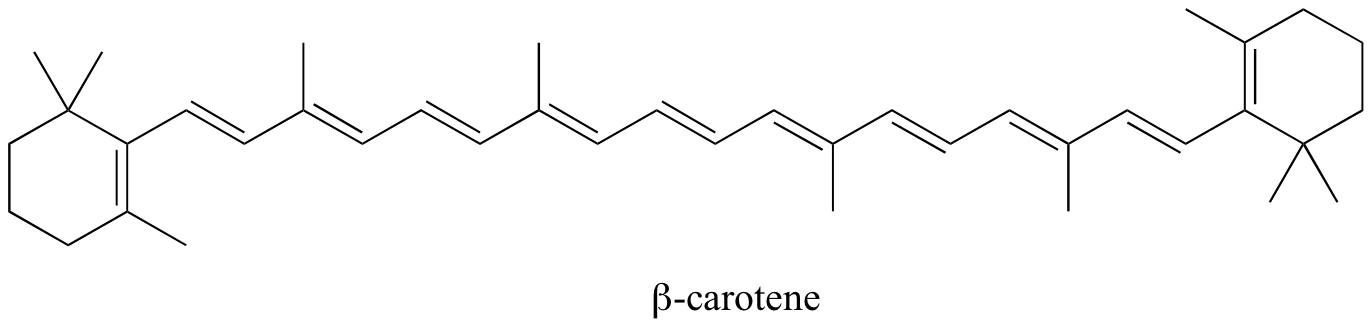
The beingness of alternating double bonds creates many `pi` and `pi^"*"` orbitals. Remember that, when atoms absorb energy, they absorb a specific wavelength of energy. In conjugated systems, the amount of free energy absorbed corresponds to the `pi` to `pi^"*"` transition. This is the same concept of emission from `n=4` to `north=1` energy levels.
This is the color that arises from organic paints or, if you use fountain pens, fountain pen inks. The reason that organic paints don't concluding long, yet, is that overtime the double bonds go abroad due to reactions with the moisture in air. The removal of this double bail removes the `pi` to `pi^"*"` transition, which removes the color.
Fun fact inside a fun fact: this is what bleach does. The reason bleach removes color is that it removes double bonds, thereby eliminating the `pi` to `pi^"*"` transition.
ii. What is special about `F_2` and `O_2` that they warrant a new MO diagram?
The answer to this question requires more agreement of orbital behavior than nosotros previously have. The curt and unproblematic (possibly unsatisfying) answer is that `F_2` and `O_2` are so electronegative that they "pull" the `sigma_"2p"` orbital closer to them. This volition exist a question we reply subsequently in inorganic chemistry, so keep an centre out until then.
Source: https://sansona.github.io/articles/mo-diagrams.html
0 Response to "How to Draw Molecular Orbital Diagram of O2"
Post a Comment